The Holy Grail of Energy
Ever wondere how the Sun could have been shining for 4.55 billion years, radiating immense amounts of energy into space? How does the Sun produce the energy that will keep it shining for another 5 billion years? The energy source that powers the Sun is a “fusion reactor” buried at its center, where the nuclear reactions convert hydrogen into helium, accompanied by the release of huge amounts of energy.
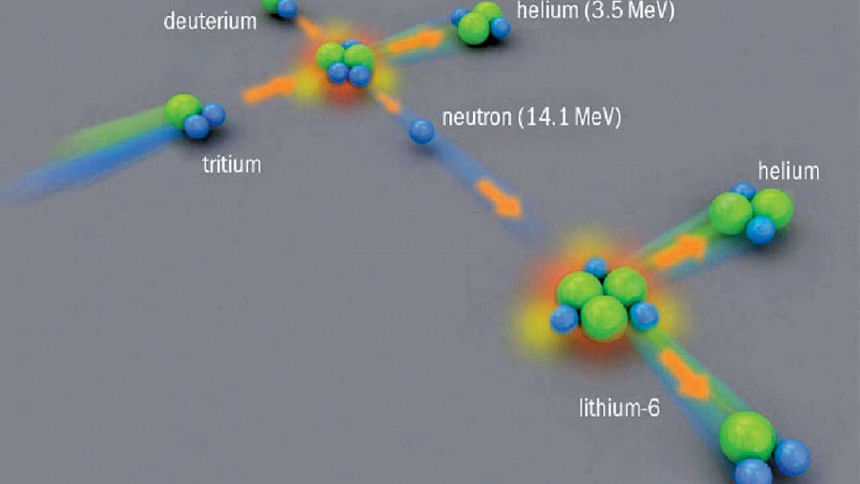
Nuclear fusion, the holy grail of energy, is a process in which two lighter nuclei are fused together to produce a heavier nucleus. In the process, a good deal of energy is released, three to four times greater than the energy released by fission reactions that are at the heart of today's nuclear power plants. The simplest reaction in which enormous amount of energy will be released is the fusion of the hydrogen isotopes deuterium and tritium producing helium and a neutron. Isotopes are different forms of a nucleus containing equal numbers of protons but different numbers of neutrons.
Deuterium is found aplenty in ocean water. The amount is prodigious (33 milligrams per liter of water), enough to last for billions of years. Naturally occurring tritium, on the other hand, is extremely rare. Nevertheless, it can be produced inside a reactor by neutron activation of lithium, the other raw material for fusion found in brines, minerals and clays. Because of the abundance of fusion fuel, the amount of energy that can be released in controlled fusion reactions is virtually unlimited.
The scientists and engineers at Lockheed working on the fusion reactor acknowledge that the project is still in its nascent stage, and many key engineering challenges have to be overcome before a viable prototype can be built. Nonetheless, they are cautiously optimistic that an operational fusion reactor with enough energy output to power a small city, yet small enough to fit on the back of a truck, can be built before the end of this decade.
There are many advantages of fusion reactors. They will produce copious amount of electric power, five times more than the amount of energy it will need to heat the fusing nuclei to the desired temperature, without effecting the environment.
Unlike fission, fusion will have a low burden of waste. The by-products of fusion reaction are helium, a harmless gas used to inflate children's balloons, and neutrons. Some radioactive wastes will be produced though, but their inventory is much less than those from fission and they will be short-lived.
A fascinating application for the abundant energy that fusion may provide is the “fusion torch,” a star-hot flame or high temperature plasma into which all waste materials—liquid sewage or solid industrial refuse—could be dumped.
Good things in life don't come cheap. Once fusion reactors become a reality, they will provide the setting for a better world in our own time.
The writer is Professor of Physics at Fordham University, New York.

 For all latest news, follow The Daily Star's Google News channel.
For all latest news, follow The Daily Star's Google News channel. 




Comments