COVID-19: What is it exactly?
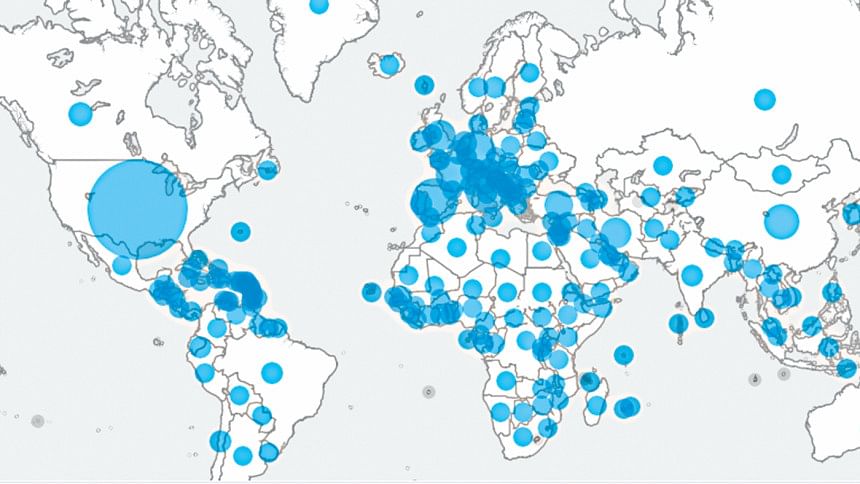
The usual pace of human life has suddenly come to a halt, thanks to an invisible organism. A novel form of coronavirus, SARS-CoV-2, responsible for the coronavirus disease (COVID-19), has completely changed our lives in just a few months. On 11 March 2020, the World Health Organization (WHO) officially declared the COVID-19 as a pandemic. The confirmed cases of COVID-19 have exceeded 1.79 million globally, with a death toll of over 109,654, according to worldometers, as of 12 April, 2020. However, the available data shows a difference in the severity of coronavirus outbreaks between the countries that reacted quickly and decisively with social distancing and preventative measures such as Taiwan, Vietnam, etc., and those that acted late such as Iran, Italy, UK, etc. Scientists all over the world are working very hard to explain why the virus is behaving in such an extreme way.

If we understand the structure of SARS-CoV-2, we might be able to understand why it has spread so rapidly. Coronaviruses are RNA viruses enveloped in a lipid bilayer – each virus particle has a small set of genes, enclosed by a sphere of fatty lipid molecules which looks like a spiky ball under the microscope. The spikes on the surface of the virus, also known as the S protein to the scientific community, stick to a protein called Angiotensin Converting Enzyme 2 (ACE2) found on the surface of certain lung cells. This is a known mechanism of the entry of coronavirus into our body. What is new in the case of SARS-CoV-2 is that it has undergone some mutations that allow the virus to interact more strongly with ACE2. Therefore, its ability to infect people has become much higher than its cousin, the classical SARS-CoV. In short, SARS-CoV-2 is internalised into our cells after binding to ACE2, remains in a compartment called endosomes inside the cells, sheds its outer protective layer utilising the slightly acidic nature of the endosomes, releases its genetic material, produces more viral particles using the host body (in this case it is us, humans), assembles the viral particles to form a new infectious virus and goes out of the cells to infect new cells and, potentially, new people. The novel SARS-CoV-2 has engineered itself to utilise the host body more efficiently that gives it replication and survival advantage. The dying lung cells are sloughed, fill the airways with junk, and the virus gets deep inside. Eventually the lungs get clogged with dead cells and fluid, which makes it extremely difficult to breathe. The virus may progress to infect ACE2-bearing cells in other organs.
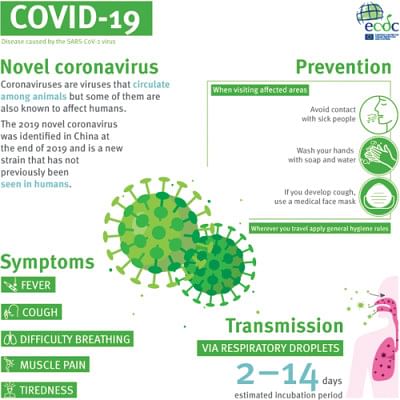
As we know, the immune system will fight back in order to get rid of the virus. This causes inflammation and fever. However, in simple terms, our immune system goes crazy in extreme situations. It causes more damage than the virus would do. Blood vessels open up, allowing immune cells to reach the site of infection. Ideally, that is what we want as a defence mechanism. However, if the blood vessels are too leaky, it will fill the lungs with even more fluids. Such an over-reactive immune response leads to a situation called cytokine storm. Cytokine storm leads to more lung damage and eventually develops acute respiratory distress syndrome (ARDS). Breathing becomes difficult, resulting in lack of oxygen in the body. This was responsible for many deaths in the 1918 Spanish flu pandemic as well as the 2003 SARS outbreak. It is highly probable that the same might be happening in case of COVID-19. During the cytokine storm, immune system loses control and goes on without hitting the right target, resulting in people becoming more susceptible to bacterial infection. In addition, organs other than lungs, such as the heart, may also be affected by the cytokine storm. This might explain why COVID-19 patients develop secondary bacterial infection and multi-organ failure.
The question is why do some people get incredibly sick with the virus, while others do not? Age could be a factor. Elderly people are thought to be more susceptible possibly because of their compromised defence system that cannot start the effective initial response. On the contrary, children are generally found to be less affected as their immune system is less likely to develop the cytokine storm. However, clinical cases suggest no one is safe; everyone is getting affected. This makes the case more complicated and brings into play genetic factors, the viral load one is exposed to, and other microorganisms present in the individual's body.

The New England Journal of Medicine has published a report on SARS-CoV-2's stability on surface and in aerosol (air). The study showed that the SARS-CoV-2 can remain stable for more than three hours in aerosol, 72 hours on plastics, 48 hours on stainless steel, 24 hours on cardboards and four hours on copper, allowing the virus to infect more readily. Let us consider R0, a parameter that shows the infectability of a virus. The R0 value of SARS-CoV-2 is around 2.5, meaning one person infects another 2.5 people on average. This is not a small number from the viewpoint of epidemiology. However, this R0 value of SARS-CoV-2 is lower than that of related diseases such as SARS and MERS. One may wonder then, what is the big deal about SARS-CoV-2? Why are people so worried?
Most respiratory viruses tend to infect either the upper or lower respiratory tract. An upper-respiratory infection spreads more easily but tends to be milder. On the other hand, a lower-respiratory infection is harder to transmit, but is more severe. However, with the case of the current SARS-CoV-2, different studies show that it seems to infect both upper and lower airways owing to their ability to exploit the host system better. This double-faced nature of this virus explains why it can spread among people before the symptoms even show up. It means the infected person starts shedding and transmitting SARS-CoV-2 while he or she is asymptomatic. This trait of the new virus has made the management and control of the disease very difficult. Perhaps it even transmits while still confined to the upper airways, before making its way deeper and causing severe symptoms. This makes the disease even more severe than related SARS (classic) and MERS diseases because the latter ones were usually transmitted after we could see the symptoms. That is, SARS-CoV-2 is mutated to become a completely different form of virus and causes a disease with different symptoms, spreads and kills more readily.

If you survive COVID-19, it is because your body has learned how to fight the virus. You have produced the antibodies against the pathogen. And if you are willing to share them, you are now someone's new best friend! On March 20, 2020 a team at Johns Hopkins University received FDA approval to try this technique. There is a high probability that it may work. This same technique was used successfully to treat Ebola in 2014. However, the approach carries a risk as well. Antibodies to a virus can make a viral infection worse in some cases, such as with dengue fever. We are yet to know how COVID-19 will react to it.
However, as of now there is no proper solution to COVID-19. The scientists' community is working hard to manage COVID-19. About 70 clinical trials are going on worldwide for developing an effective therapy, including drugs and vaccines. According to WHO, vaccination is one of the most effective ways to prevent diseases. A vaccine helps the body's immune system produce antibodies, a special protein (immunoglobulin) to recognise and fight pathogens like viruses or bacteria, which then keeps us safe from the diseases they cause. Some people may be wondering why it is taking so much time to develop "just" a vaccine. There are three basic steps: vaccine development, testing for its efficacy in human trials, and finally formulating the vaccines in marketable forms. Among these three steps, the first one (development of vaccine) takes the least amount of time, and we are still in that first step! If everything works out, we may have a vaccine for COVID-19 in 9 to18 months. On 30 March 2020, Johnson & Johnson (NYSE: JNJ) (the Company) announced the selection of a lead COVID-19 vaccine candidate from constructs it has been working on since January 2020; the significant expansion of the existing partnership between the Janssen Pharmaceutical Companies of Johnson & Johnson and the Biomedical Advanced Research and Development Authority (BARDA); and the rapid scaling of the Company's manufacturing capacity with the goal of providing global supply of more than one billion doses of a vaccine. The Company expects to initiate human clinical studies of its lead vaccine candidate at the latest by September 2020 and anticipates the first batches of a COVID-19 vaccine could be available for emergency use authorisation in early 2021, a substantially accelerated timeframe in comparison to the typical vaccine development process.

When it comes to drugs that can be used in treating patients with this virus, a recent clinical trial conducted in France indicated that hydroxychloroquine (an anti-malarial drug) in combination with azithromycin (an antibacterial agent) might be an effective therapeutic strategy. FDA has given emergency-use authorisation for hydroxychloroquine sulfate and chloroquine phosphate to treat COVID-19 patients. Hydroxychloroquine hampers the virus' ability to replicate in two ways. Firstly, the drug enters the endosomes within the cells. Endosomes are usually acidic which is necessary for the replication of many viruses including SARS-CoV. The chemical property of the drug increases the pH of the endosomes, making them more basic in nature and thus inhibits viral replication. Secondly, the drug also prevents SARS-CoV from sitting onto ACE2 protein described earlier and hampers the entry of the virus. This stops the ability of the virus to infect. FDA is now undertaking a larger clinical trial of hydroxychloroquine because the reported clinical trial in France was carried out in a "small" cohort. Azithromycin (an antibiotic) was also used in the clinical trial to inhibit any potential secondary infection caused by bacteria in SARS-CoV-2-infected patients. Some studies are also using zinc (Zn), a trace element, and people wonder why. Zn inhibits RNA-dependent RNA polymerase (RdRP) and blocks viral replication. But Zn cannot enter the cell by itself. It requires an ionophore (a carrier) to get inside the cell, such as chloroquine group of drugs. It should be noted that there is currently no FDA approved vaccine or drug for COVID-19. However, antiviral drugs used in influenza such as Favipiravir and Remdesivir and a combination of anti-HIV drugs Lopinavir-Ritonavir are being reported to be promising drug candidates in the treatment of COVID-19. Favipiravir, a patented drug in Japan, has been recently designated for Japan's standard treatment for COVID-19. China has also previously announced the use of Favipiravir to be safe and effective in treating COVID-19 patients.
Although corticosteroid (anti-inflammatory drug) treatment is not routinely recommended to treat SARS-CoV-related pneumonia, according to the new pathological findings of pulmonary edema and hyaline membrane formation, timely and appropriate use of corticosteroids together with ventilator support may be considered for the severe COVID-19 patients to prevent acute respiratory distress syndrome (ARDS) development, which is thought to be majorly associated with COVID-19 related mortality. In fact, China approved Tocilizumab, an anti-inflammatory drug, to manage their COVID-19 patients. It sounds contradictory since WHO has initially recommended not using ibuprofen, a non-steroidal anti-inflammatory drug since it has been reported to increase ACE2 production, which is beneficial for the virus since, as we discussed, COVID-19 binds with ACE2 and then enters the body. Thus, in this case ibuprofen's action is not desired. WHO has also recommended discontinuing Captopril (Angiotensin Converting Enzyme, ACE, inhibitors) and Valsartan (Angiotensin Receptor Blockers, ARBs) classes of drugs in hypertensive patients infected with COVID-19. This recommendation was based on animal data. The European Society of Cardiology, however, has highly encouraged the continuation of these drugs in hypertensive patients until enough human data is available to prove the benefits of their discontinuation. The reason is again underlying molecular mechanism. It has been shown in separate studies that discontinuing ACE inhibitors and/or ARBs would, in fact, increase the free ACE2 level in the lungs, which is used by COVID-19 to enter the body. Therefore, discontinuing these drugs may worsen the COVID-19 infection. However, all these studies must be validated before we get an effective therapeutic regimen.
So, the question is, what do we do? Ideally, we need a vaccine that is effective and can be produced in mass. It might take several months. Until we have a vaccine against the novel SARS-CoV-2, self-quarantining is the only measure to be taken to prevent the sharp rise of the viral infection, though this is not easy. "Flattening the curve" also requires extensive monitoring of each individual to keep the infected people away from transmitting the virus further. That is exactly how Singapore and Taiwan initially managed to control the further spread of this deadly disease. We must consult with the health care providers and follow the guidelines provided by the Institute of Epidemiology, Disease Control and Research (IEDCR). Health care providers can best advise what to do if one has symptoms of COVID-19. For now, the only short-term solution is social distancing and maintaining good hygiene. Social distancing means complete physical isolation and not visiting your loved ones so that you do not put them in a more dangerous situation than yourself. Good hygiene includes washing our hands frequently and properly with soap and water or with an alcohol-based hand sanitiser. The lipid shell of the virus gets ripped apart by soap in 20 seconds of thorough handwashing. If soap and water are not available, using hand rubs is considered to be effective since it destroys the virus. Surfaces also need to be cleaned with disinfectants. FDA has also just announced that devices intended to kill pathogens or microorganisms in the air, such as sterilisers, disinfectant devices, air purifiers, etc., need to be made available which will assist removing this deadly virus from the environment. These are the right things to do to fight COVID-19.
The COVID-19 situation has escalated into an unprecedented global crisis in just a few weeks. It has impacted everyone. Each of us can help stop the spread of COVID-19 by maintaining strict social distancing and good hygiene. Please don't panic. We need to be aware and vigilant to fight this virus.
Ridwan Islam is a Senior Lecturer (on study leave) of the Department of Pharmacy, BRAC University. He is currently a PhD Candidate at the University of Nebraska, Medical Center, USA.
Professor Dr Eva Rahman Kabir is the Head of the Department of Pharmacy, BRAC University and Former Professor, Department of Pharmaceutical Technology, University of Dhaka.
Dr Hasina Yasmin is a Professor of the Department of Pharmacy, BRAC University.

 For all latest news, follow The Daily Star's Google News channel.
For all latest news, follow The Daily Star's Google News channel. 



Comments