Cutting Edge Genomic Research in Bangladesh
Few contemporary advances in biotechnology have captured public imagination as much as the discovery of the CRISPR-Cas system. Startups like Intellia Therapeutics and Editas Medicine have raised hundreds of millions of dollars on the promise of CRISPR-based gene editing. This could be a truly revolutionary moment in the history of human technology.
But what exactly is the CRISPR-Cas system? CRISPR-Cas is a collection of DNA sequences and proteins often found in bacteria that protects them from viral infection. It can act as a kind of bacterial “immune system”—recognising very specific snippets of viral DNA, and then destroying these DNA sequences to eliminate invading viruses. Just as human beings are engaged in a constant immune war against infectious bacteria and viruses, bacteria themselves struggle against viruses that attack and kill them. These bacteria-killing viruses are called bacteriophages—“eaters of bacteria”.
It is the ability of the CRISPR-Cas to target and destroy very specific DNA sequences that makes it a powerful tool for genetic engineering. If the system was not so precise the bacteria could miss its target, or risk destroying its own DNA and killing itself. The same precision can now be used by genetic engineers—in 2017 CRISPR gene editing was used to cure genetic deafness in mice, and it is only a matter of time before similar applications are extended to human patients.

As with many tools in biotechnology, from painkillers to vaccines, the CRISPR-Cas system was not synthesised from scratch by human engineers and chemists. It is something already found in the wild, a product of nature that we can reshape for our own purposes. Thus, to fully realise the potential of technologies like CRISPR gene editing we must continue to generate more knowledge on the natural diversity and functionality of this system.
Many people would be surprised to hear that such a cutting-edge topic in biology is an active area of research and discovery in Bangladesh, but this is exactly the case with recent research conducted at icddr,b by Professor Shah Faruque and colleagues. Professor Faruque had maintained an active research team in icddr,b for over two decades, and has now moved to BRAC University to strengthen life science research in the Department of Mathematics and Natural Sciences.
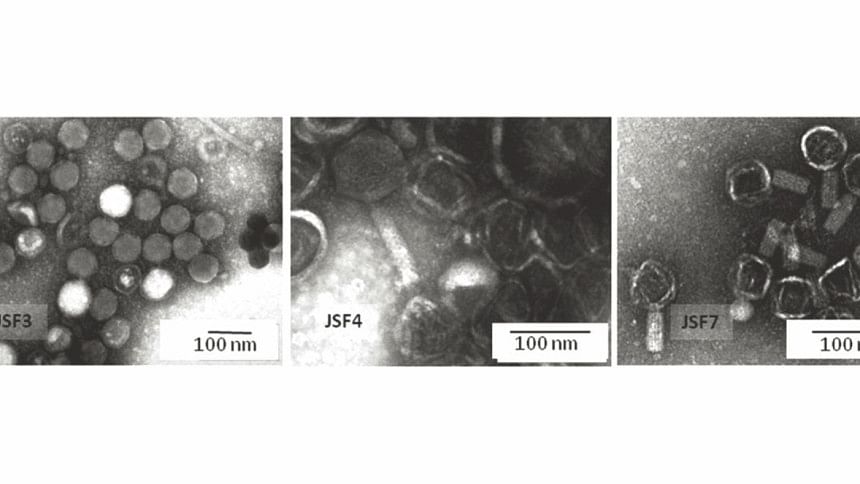
Professor Faruque's findings, recently published in the prominent Nature Publishing Group journal Scientific Reports, analyse the CRISPR-Cas system in a group of cholera bacteriophages, viruses that attack Vibrio cholerae, the bacteria responsible for causing cholera. Dr Faruque's results build on the research of Andrew Camilli, who discovered these viral CRISPR-Cas elements in 2013.
The vast majority of CRISPR-Cas systems studied previously exist in bacteria to defend against viruses. In a remarkable twist, the particular elements studied by Professor Faruque exist instead in the viruses that attack bacteria.
These cholera-killing viruses are our allies in the fight against this deadly diarrhoeal disease—previous work by Professor Faruque's group has shown that these naturally occurring viruses help to stop seasonal cholera epidemics in Bangladesh by killing the pathogenic bacteria in contaminated water.
Professor Faruque's recent paper uses cutting-edge whole genome sequencing to show that these viruses use the CRISPR-Cas system to suppress the defences of cholera bacteria and infect them more effectively. By studying DNA sequences from multiple viruses and bacteria collected in Bangladesh over many years, Dr Faruque and his group were also able to demonstrate that the viral CRISPR-Cas system is continuously evolving. The virus and the bacteria are locked in an “evolutionary arms race”, their genetic material constantly mutating to counter the other's defence mechanisms.
These findings could be used to engineer viruses to kill the cholera pathogen for therapy or for environmental control of deadly epidemics. Given the serious danger that cholera epidemics pose during times of crisis in developing countries, from the conflict in Yemen to the crowded camps of displaced Rohingya Myanmar nationals in Bangladesh, this work could have great value for saving human life in the future.
Perhaps the most remarkable aspect of this research is that it was entirely done by Bangladeshi scientists, with no foreign collaborators. These results are a testament to the value of supporting local scientists and developing our own research capacity, without being overly reliant on foreign specialists. Other members in Professor Faruque's research team in icddr,b who contributed substantially to this work are Iftekhar Bin Naser, M Mozammel Hoque, M Ausrafuggaman Nahid, Tokee M Tareq and Kamruzzaman Rocky.
Professor Faruque has recently left his position at icddr,b to start a new research group at BRAC University, where he can focus on expanding the research capability of one of the nation's premier private universities. When asked about his future plans at BRAC University, Professor Faruque expresses his motivations for this new initiative with passionate clarity: “The purpose of this endeavour is to build the life science research capabilities of a Bangladeshi university, to increase its visibility to the donor community, and in turn attract more research grants, to be able to build the next generation of Bangladeshi researchers.”
Zain Omar Ali is a freelance science writer and received his Master's in Molecular and Cellular Biology from Harvard University.

 For all latest news, follow The Daily Star's Google News channel.
For all latest news, follow The Daily Star's Google News channel. 




Comments